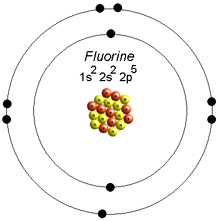
The lowest value is assigned to the shell located closest to the nucleus while the shell farthest from the nucleus has the highest value. The Bohr model names these shells as K, L, M, N, etc., or 1, 2, 3, 4, etc. Each electron has its specific energy level and each energy level has a specific number of electrons. These are also known as orbits or energy levels. Shells: The definite circular path followed by the electrons, inside an atom, around the nucleus. 
The electrons follow a definite path while revolving around the nucleus.
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
